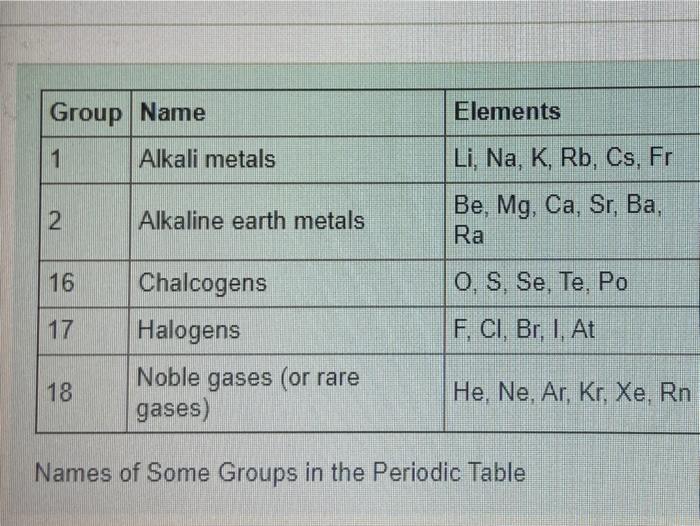
Size of Atoms - Trends Atomic Radii Li Na K Rb Cs Cl S P Si Al Br Se As Ge Ga I Te SbSn In Tl Pb Bi Mg Ca Sr
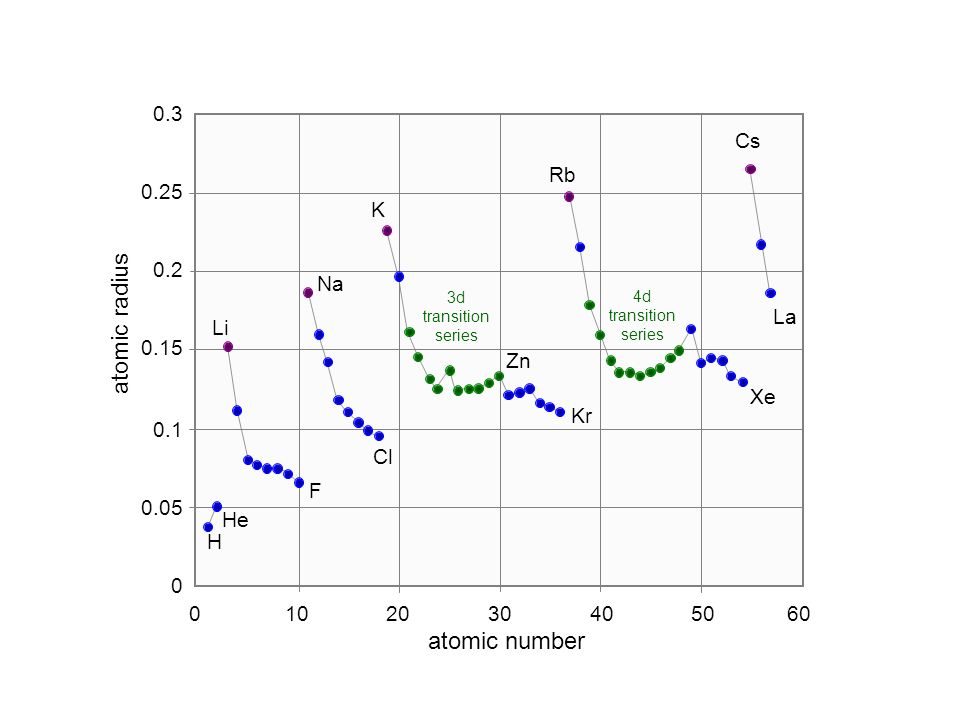
Size of Atoms - Trends Atomic Radii Li Na K Rb Cs Cl S P Si Al Br Se As Ge Ga I Te SbSn In Tl Pb Bi Mg Ca Sr
![Alkali Metal (Li, Na, K, Rb, Cs) Mediation in Magnesium Hexamethyldisilazide [Mg(HMDS)2] Catalysed Transfer Hydrogenation of Alkenes - Gentner - 2021 - ChemCatChem - Wiley Online Library Alkali Metal (Li, Na, K, Rb, Cs) Mediation in Magnesium Hexamethyldisilazide [Mg(HMDS)2] Catalysed Transfer Hydrogenation of Alkenes - Gentner - 2021 - ChemCatChem - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/9475fd99-b13c-401a-92d2-68e7b4b978ed/cctc202100218-fig-5002-m.jpg)
Alkali Metal (Li, Na, K, Rb, Cs) Mediation in Magnesium Hexamethyldisilazide [Mg(HMDS)2] Catalysed Transfer Hydrogenation of Alkenes - Gentner - 2021 - ChemCatChem - Wiley Online Library

Moar conductivites of Li^(+),Na^(+),K^(+) "and" Rb^(+) ions in aqueous solutions are in the following order.

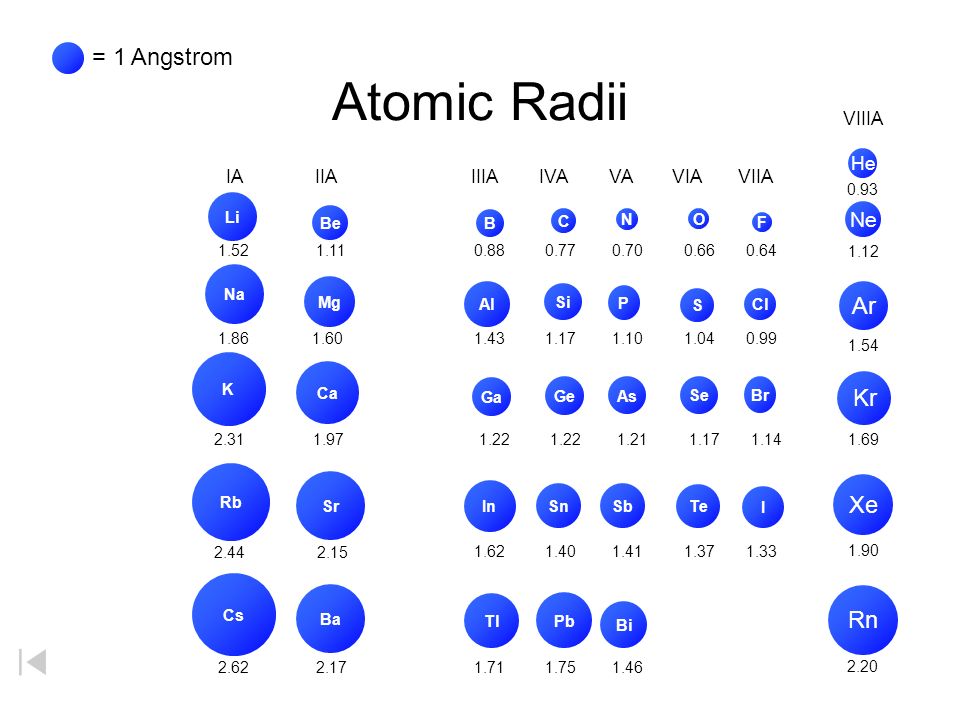
Size of Atoms - Trends Atomic Radii Li Na K Rb Cs Cl S P Si Al Br Se As Ge Ga I Te SbSn In Tl Pb Bi Mg Ca Sr

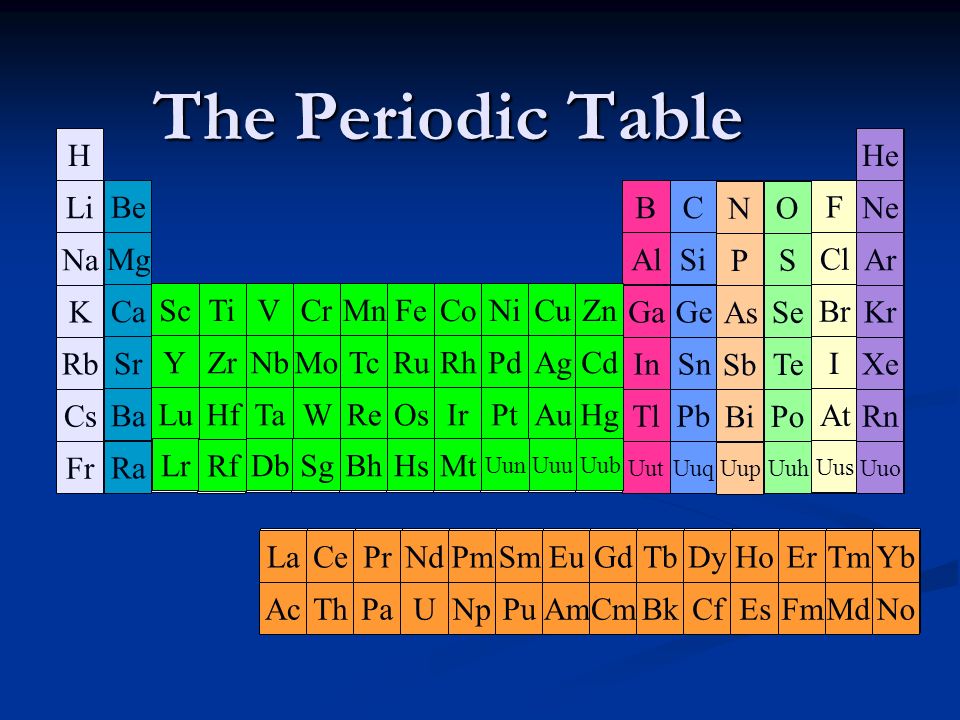
H Li Na K Rb Cs Fr Be ScTi Mg VCrMnFeCoNiCuZnGaGeSeBrCaKr YZrNbMoTcRuPdAgCdInSnSbSrTeRh BaHfTaWReOsIrAuHgTlPbBiPoLaAtPt RaRfDbSgBhHsMt??Ac? AlP NO SCl. - ppt download

Unsur Li, Na, K, Rb, Cs, Fr merupakan unsur alkali ( golongan IA)..?benar atau salah...? - Brainly.co.id

Phase Segregation in Cs-, Rb- and K-Doped Mixed-Cation (MA)x(FA)1–xPbI3 Hybrid Perovskites from Solid-State NMR | Journal of the American Chemical Society
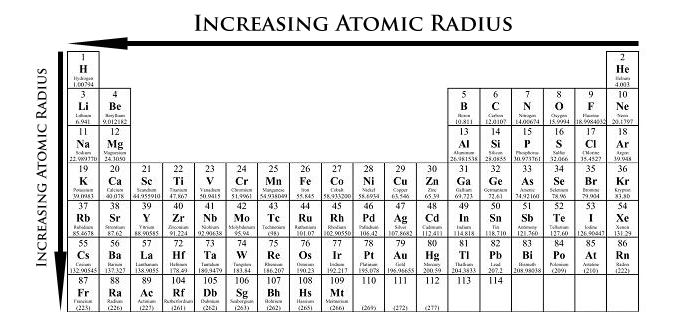
Increasing order of atomic radiiLi, Na, K, Rb, Cs:(A) Li Na K Rb Cs(B) Li Na K Cs Rb(C) Li K Na Rb Cs(D) None of these
The K/Rb Ratio and Its Geological and Petrophysical Implications: A Test Case of the San Jorge Gulf Basin, Argentina; #41644 (20



![Case Based MCQ] Metallic Character: The ability of an atom to donate Case Based MCQ] Metallic Character: The ability of an atom to donate](https://d1avenlh0i1xmr.cloudfront.net/393fca25-8983-4d07-b323-91177250cc46/question-5-cased-based-chapter-11---teachoo.jpg)